|
|
Dynamic Chiropractic August 2018
Low back pain will affect some 80 percent of all adult Americans at some point.(1) However, it’s not just an American problem. Low back pain is becoming more prevalent around the world. Worldwide, disability from low back pain has risen by more than 50 percent since 1990.(2) As low back pain becomes a more international problem, the costs to patients, health care systems, and society escalates. In the U.S. alone, total costs associated with LBP exceed $100 billion per year, two-thirds of which are a result of lost wages and reduced productivity.(3)
Drugs Don’t Help
Traditional medical treatment of low back pain often involves prescription painkillers, particularly opioids. The risk of abuse and addiction with these drugs is well known. In 2016, the CDC issued revised prescribing guidelines that recommend sharply cutting the duration of any pain-killer prescription.(4) Most importantly, these drugs fail to be effective in the long term. A 2016 meta-analysis of 20 studies of opioids for low back pain, published in JAMA Internal Medicine, found that opioid analgesics for chronic low back pain provided only modest short-term pain relief, and only for some patients. In many of the studies analyzed, more than half of the participants in the trial dropped out due to adverse side effects of these dangerous drugs. None of the studies showed clinically important pain relief (defined as more than 20 points improvement on a 100-point scale). Those who did get some relief from pain took doses that were much higher than recommended. The meta-analysis brought out an important point about opioid studies: almost all are short term. Very few high-quality studies have looked at the long-term effects of opioid use for patients with chronic low back pain. A recent study that did look at a 12-month period found that treatment with opioids was no better than treatment with nonopioid medications such as ibuprofen for improving pain-related function.(5) In addition to offering little help to patients, opioid drugs can make chronic pain worse. For some patients, using opioids can induce hyperalgesia, or a paradoxical response whereby patients who take these drugs become more sensitive to painful stimuli. The mechanisms behind hyperalgesia are still unclear, but the risk that painkilling drugs could make a patient more sensitive to pain is clear.6 Hyperalgesia is another potential harm to add to the long list of bad side effects of opioid use.
Better Solutions
The evidence is very clear: opioids not only aren’t a solution to low back pain, they’re harmful to patients and may make the pain worse. Nondrug treatments, such as nonthermal low-level laser therapy led by chiropractors, are far preferable. They’re safe, they’re supported by research, they work, and they’re cost effective.
A Breakthrough Tool For Chronic Low Back Pain
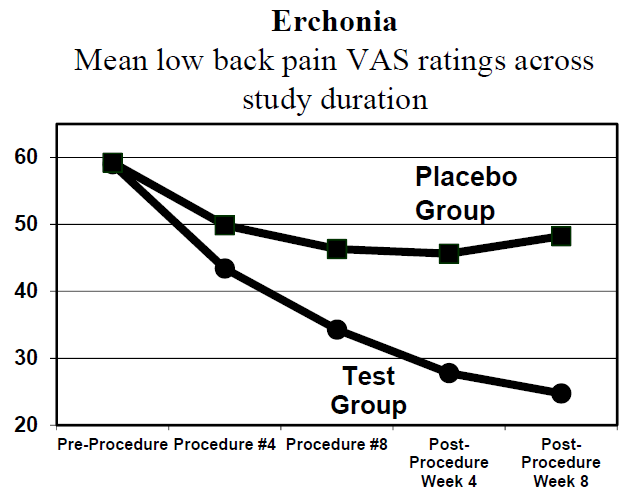
In July 2018, the Erchonia FX 635 low-level laser received official FDA clearance for relief of chronic low back pain—the first and only laser to be cleared for this purpose. Chiropractors now have a powerful new tool to help their patients relieve chronic low back pain and get back to work and normal activity. Founded in 1996, Erchonia is the first company in the world to gain an FDA market clearance for the use of low level laser therapy. The company has 16 FDA 510(k) approvals for their equipment and more Level 1 clinical trials than all other therapeutic laser manufacturers combined. The clinical trial conducted to obtain FDA clearance was designed to test the effectiveness of the Erchonia FX-635 in providing relief of chronic low back pain of musculoskeletal origin. The study was a placebo-controlled, randomized, double-blind parallel group multi-center design. Fifty-eight people completed the study; half were treated with the laser and half were treated with a placebo (sham laser 635nm LED). All the participants rated their pain as 40 or greater on the 0 to 100 Visual Analog Pain Scale (VAS). The average duration of low back pain for the participants was 97.8 months (approximately eight years). The majority of the participants had had bilateral low back pain with an average pain rating of 59.10 on the VAS.
All the participants received eight 20-minute procedure administrations across the lower back region with the Erchonia FX-635 laser (active or sham) across a four-week period: two procedures per week, each procedure three to four days apart. Of those who received the active laser treatment, 72.4 percent attained a 30 percent or greater decrease in chronic low back pain VAS rating from baseline to endpoint (see chart). Chart 1 shows the response to laser treatment among the test group and those who received placebo treatment (635 nm LED, not a laser). Among the actual treatment group, VAS ratings dropped precipitously from 59 to 23 over eight weeks. Among the placebo group, the drop was only about 10 points and the improvement quickly leveled out. No adverse events were reported during the study duration. At the end of three months, the reduction in pain was sustained, even though no additional treatment was administered after the first month.7 Low level laser treatment (LLLT) works by stimulating cell activation processes which, in turn, intensify physiologic activity. The light energy from the laser initiates a cascade of reactions that stimulate the mitochondria to increase the production of ATP. The laser light stimulates the release of natural healing chemicals that lead to rapid cell growth, increased metabolic activity, increased angiogenesis, improved vascular activity, suppression of the COX-2 inflammatory pathway, and decreased inflammation. The specific wavelength (color) of the laser light is responsible for influencing the biochemical cascades. Research has shown that a wavelength of 635 nm is ideal for accelerating healing and reducing post-injury inflammation. The coherent, pulsed light delivered by the Erchonia laser insures deep tissue stimulation and absorption. Laser treatment is painless. There’s no sensation of heat and the skin and tissue aren’t damaged in any way. The patient lies prone; the laser light is provided by the FX-635 using three diodes on an adjustable stand. The laser light is delivered to the lower back area in a moving circular pattern for twenty minutes. The patient does not need to be attended during this time. The clearance of the FX-635 is a watershed moment for the chiropractic profession for treating low back pain. (Erchonia Corporation www.Erchonia.com)
|
| Rob Silverman, DC, DABCO
https://www.drrobertsilverman.com/ REFERENCES: 1. Lemeunier, N, Leboeuf-Yde, C, and Gagey, O. The natural course of low back pain: a systematic critical literature review. Chiropract Man Ther. 2012; 20: 33 2. Stephanie Clark, Richard Horton. Low back pain: a major global challenge. The Lancet. Volume 391, No. 10137, p2302, 9 June 2018. DOI: https://doi.org /10.1016/S0140- 6736(18)30725-6 | 3. William Thomas Crow, DO; David R. Willis, DO, MBA. Estimating Cost of Care for Patients with Acute Low Back Pain: A Retrospective Review of Patient Records. The Journal of the American Osteopathic Association, April 2009, Vol. 109, 229-233. 4. Deborah Dowell, MD; Tamara M. Haegerich, PhD; Roger Chou, MD. Morbidity and Mortality Weekly Report (MMWR), CDC Guideline for Prescribing Opioids for Chronic Pain — United States, 2016; March 18, 2016, 65(1);1–49. 5. Krebs EE et al. Effect of Opioid vs Nonopioid Medications on Pain-Related Function in Patients with Chronic Back Pain or Hip or Knee Osteoarthritis Pain: The SPACE Randomized Clinical Trial. JAMA. 2018 Mar 6;319(9):872-882. doi: 10.1001/ jama.2018.0899. 6. Lee M et al. A comprehensive review of opioid-induced hyperalgesia. Pain Physician. 2011 Mar-Apr;14(2):145-61. 7. Market Clearance to Treat Chronic Low Back Pain.(FX 635) – 2018 – A Placebo-Controlled, Randomized, Double-Blind, Parallel-Group, Multi-Center
|